Health Advice
/Health

Power outages linked to heat and storms are rising, and low-income communities are most at risk, as a new NYC study shows
Many Americans think of power outages as infrequent inconveniences, but that’s quickly changing. Nationwide, major power outages have increased tenfold since 1980, largely because of an aging electrical grid and damage sustained from severe storms as the planet warms.
At the same time, electricity demand is rising as the population ...Read more
Baby orangutan born at Tampa's Busch Gardens via C-section was a rare feat
TAMPA, Fla. — Luna loved the ultrasound game.
The 26-year-old orangutan at Busch Gardens would come up to the mesh screening that separates the primates from the humans that care for them and happily present her belly.
Dr. Maria Spriggs, chief veterinarian at Busch Gardens, would use that play time to put an ultrasound probe on Luna’s ...Read more

Environmental Nutrition: Spring into asparagus
Asparagus is a seasonal favorite.
The folklore
Asparagus, whose name means stalk or shoot in Greek, was first cultivated about 2,500 years ago in Greece, where it was used medicinally to treat toothaches and help prevent bee stings. Considered a delicacy since ancient times, this dainty and nutritious spear continues to be a delicious ...Read more

5 healthy reasons to crave more cauliflower
The goodness of cauliflower goes beyond its ability to become almost anything you can imagine in the kitchen — a crust, a “wing,” a rice, a mash, and the list goes on. Cauliflower’s mild flavor, satisfying texture, and chameleon-like versatility makes it not only a hearty and nutritious add-in as is, but also an inventive and savvy swap ...Read more

Cellulitis: How long does it take to heal on legs?
Cellulitis is an infection of the deep layers of the skin. It develops when bacteria enter through a cut, bite, or wound — including tiny breaks in cracked, dry skin. Common skin-dwelling bacteria, Staphylococcus or Streptococcus, are the usual culprits. Although cellulitis can occur anywhere on the body, the most common location is the lower ...Read more

Mayo Clinic Q&A: What is cholangiocarcinoma and how is it treated?
DEAR MAYO CLINIC: My mother has been experiencing unusual and persistent fatigue, abdominal pain, and jaundice. After undergoing a CT scan, her doctor diagnosed her with cholangiocarcinoma. What is this type of cancer? And what treatment options are available?
ANSWER: Cholangiocarcinoma is a rare cancer that develops from the bile ducts, which ...Read more

Mayo Clinic Minute: Using lasers on the brain to treat seizures
For roughly a third of people with epilepsy, medication does not control their seizures. Depending on where those seizures originate in the brain, laser therapy can be an option for adults and children.
It's treatment for epileptic seizures that uses a laser on the brain.
"LITT is laser interstitial thermal therapy," says Dr. Jamie Van Gompel,...Read more

Exposed to Agent Orange at US bases, veterans face cancer without VA compensation
As a young GI at Fort Ord in Monterey County, California, Dean Osborn spent much of his time in the oceanside woodlands, training on soil and guzzling water from streams and aquifers now known to be contaminated with cancer-causing pollutants.
“They were marching the snot out of us,” he said, recalling his year and a half stationed on the ...Read more
Mayo Clinic Minute: Reasons to be a living kidney donor
Nearly 100,000 people in the U.S. are on the waiting list for kidney transplant, according to the Organ Procurement and Transplantation Network. Finding a donor kidney that is compatible and matches the right blood and tissue types can be the biggest challenge, especially when the donor pool isn't as large as the number of people who need a ...Read more

Mayo Clinic Minute: Good carbs for optimal health
Do you push away the breadbasket or opt out of any meal that includes macaroni? If so, chances are you are one of the millions of people watching your carbs, or carbohydrates.
Dr. Christine Nguyen, a Mayo Clinic family physician, says not all carbs are bad, and there are plenty of foods with good carbs. Fiber-rich foods like whole grains, ...Read more

‘It’s a deep emotional ride’ – 12 young people in Philly’s toughest neighborhoods explain how violence disrupts their physical and mental health
In 2023, 410 people were murdered in Philadelphia – more than a quarter of them under age 25. In addition to the people who died, countless others lost loved ones and people they relied on.
As a social scientist who studies different forms of violence and how they affect a person’s health, I know that the violence young people in ...Read more
Cancer patient raises funds for research, provides support for others
ATLANTA — As Kim Airhart battles her own cancer recurrence, she continues to raise funds and awareness for ovarian cancer research and provide support for other women fighting the harrowing disease.
Statistically, patients diagnosed with Stage 4 ovarian cancer have a high rate of recurrence, but Airhart hoped she would be a unicorn. Through ...Read more

US long-term care costs are sky-high, but Washington state’s new way to help pay for them could be nixed
If you needed long-term care, could you afford it?
For many Americans, especially those with a middle-class income and little savings, the answer to that question is absolutely not.
Nursing homes charge somewhere around US$100,000 a year, while frequent visits from a paid caregiver may set you back more than $5,000 a month. ...Read more

To stop fentanyl deaths in Philadelphia, knocking on doors and handing out overdose kits
PHILADELPHIA -- On a narrow street lined with row houses and an auto body shop in the Kensington neighborhood of North Philadelphia, Marsella Elie climbs a home’s front steps and knocks hard on the door.
A middle-aged man appears with a wary look on his face.
“Hello, sir, how are you doing today?” asked Elie, wearing a royal-blue jacket ...Read more

What Florida's new 6-week abortion ban means for the South, and traveling patients
Monica Kelly was thrilled to learn she was expecting her second child.
The Tennessee mother was around 13 weeks pregnant when, according to a lawsuit filed against the state of Tennessee, doctors gave her the devastating news that her baby had Patau syndrome.
The genetic disorder causes serious developmental defects and often results in ...Read more
Mayo Clinic Minute: Treatment and research of glioblastoma
Considered the most aggressive type of brain cancer, glioblastoma grows rapidly, destroying healthy tissue. Globally, approximately 300,000 people are diagnosed each year with glioblastoma.
Glioblastoma is a type of cancer affecting glial cells, which connect nerve cells and support brain function.
"It tends to be a tumor that also tends to ...Read more
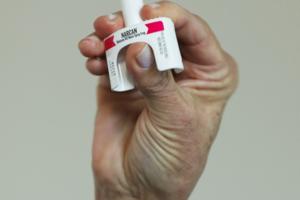
California strikes deal for cheaper overdose-reversing medication
California struck up a new deal with Amneal Pharmaceuticals on Monday to bolster the state’s supply of the opioid overdose-reversing medication naloxone at a much lower price for schools, police departments and others trying to ease the toll of fentanyl.
The deal would put the price tag for a two-dosage pack of FDA approved naloxone, often ...Read more
North Carolina health plan discriminates against transgender workers, federal appeals court rules
CHARLOTTE, N.C. — A divided federal appeals court ruled that North Carolina’s state health insurance plan discriminated against transgender patients by not covering gender-affirming care.
Transgender state workers sued the state in 2019 over a coverage exclusion in the State Health Plan for treatments for gender dysphoria, the medical term ...Read more

How LA County is trying to remake addiction treatment -- no more 'business as usual'
LOS ANGELES -- Gary Horejsi wrestled with the decision before him, knowing a life could be in his hands.
It was the third time that the woman had used drugs or alcohol since coming to CRI-Help, which runs a 135-bed residential facility in North Hollywood where people are treated for substance use disorder.
CRI-Help needed to be a safe place ...Read more

Ask the Pediatrician: How to help your child avoid the pitfalls of perfectionism
Every parent wants their child to be successful in life. But young people sometimes set excessively high standards for themselves. If something they do isn't flawless, they may become overly self-critical. Their pursuit of perfection can become unhealthy and actually interfere with what they want to accomplish.
As adolescents face the ...Read more
Popular Stories
- US long-term care costs are sky-high, but Washington state’s new way to help pay for them could be nixed
- Cellulitis: How long does it take to heal on legs?
- Mayo Clinic Q&A: What is cholangiocarcinoma and how is it treated?
- 5 healthy reasons to crave more cauliflower
- Mayo Clinic Minute: Good carbs for optimal health





