Health Advice
/Health

Environmental Nutrition: Lime time!
Tart and tangy, limes are the key ingredient that make flavors pop in our favorite sweet and savory foods.
The folklore
This tiny citrus fruit can certainly steal the spotlight as the star in tasty key lime pie, limeade and margaritas, but it shines even brighter in supporting roles, lending brightness and zing as condiment, garnish or finish....Read more

6 healthy summer foods to add to your diet
It’s almost summer — that amazing time of year when fresh produce abounds. We love that there’s an abundance of fresh, delicious and healthy choices. Better yet, many of summer’s fruits and vegetables are brimming with secret health benefits. Here are some of our favorites and why they're a particularly good choice in the summer.
1. ...Read more

What does a birth doula do?
Childbirth — painful, messy, unpredictable — has been part of humankind for time immemorial. And in the U.S., which has surprisingly high rates of avoidable complications and maternal deaths, more people seem to be seeking out doulas for additional care during pregnancy and birth, says Natalia Richey, interim chief midwife in the Department ...Read more

Mayo Clinic Q&A: How can hospice care provide comfort to those with terminal illnesses?
DEAR MAYO CLINIC: I have a loved one who was referred for hospice care. I’m not sure what this means. Can you share more about what hospice care is and how it may help my family member?
ANSWER: Hospice care might be an option for people who are nearing the end of life due to a terminal illness and have exhausted all other treatment options.
...Read more
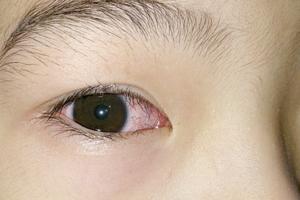
Mayo Clinic Q & A: Seeing clearly: Understanding pink eye
DEAR MAYO CLINIC: My entire family was recently affected by pink eye, including my mom, my nephews, my sister and even her dog! How is it so contagious? And what is it?
ANSWER: Pink eye is an inflammation of the lining of the eyelid and eyeball. The medical term for pink eye is conjunctivitis. Just as its name, pink eye, tells us, the most ...Read more

Mayo Clinic Q&A: Managing migraine: What's right for me?
DEAR MAYO CLINIC: I've dealt with migraine attacks for years. So has my mom. I typically manage by using over-the-counter pain medication that includes caffeine as soon as my symptoms begin. Then, if my migraine doesn't improve, I use a prescription triptan medication. But my headache days have been increasing, and my doctor suggested I try a ...Read more

Do dying people have a 'right to try' magic mushrooms? 9th Circuit weighs case
Do dying patients have a "right to try" illegal drugs such as psilocybin and MDMA if they might alleviate end-of-life suffering from anxiety and depression?
That question is now before one of the nation's highest courts, with a Seattle-based palliative care physician appealing a U.S. Drug Enforcement Administration decision barring him from ...Read more

California is investing $500M in therapy apps for youth. Advocates fear it won't pay off
With little pomp, California launched two apps at the start of the year offering free behavioral health services to youths to help them cope with everything from living with anxiety to body acceptance.
Through their phones, young people and some caregivers can meet BrightLife Kids and Soluna coaches, some who specialize in peer support or ...Read more

Bird flu is bad for poultry and dairy cows. It's not a dire threat for most of us -- yet
Headlines are flying after the Department of Agriculture confirmed that the H5N1 bird flu virus has infected dairy cows around the country. Tests have detected the virus among cattle in nine states, mainly in Texas and New Mexico, and most recently in Colorado, said Nirav Shah, principal deputy director at the Centers for Disease Control and ...Read more
Lung cancer is the deadliest of all cancers, and screening could save many lives − if more people could access it
Many medical organizations have been recommending lung cancer screening for decades for those at high risk of developing the disease.
But in 2022, less than 6% of people in the U.S. eligible for screening actually got screened. Compared with other common cancer screenings, lung cancer screening rates fall terribly behind. For ...Read more

Mayo Clinic Q & A: Going plant-based: Exploring the myths, health benefits
DEAR MAYO CLINIC: More and more of my friends are following the plant-based diet trend. Is that the same as going vegan? Is it healthy for you? What can I actually eat?
ANSWER: Plant-based foods go beyond a "boring salad." With a whole-food plant-based diet, we focus on eating foods that are primarily coming from plants and that occur in their ...Read more

One day in the frantic life of a children's emergency room nurse
It's not exactly a superpower, but nurse Joe Schwartz can tell with his nose what is causing his patient in the Children's hospital emergency department in St. Paul to look so weak, so bony thin.
When 10-year-old Juliana Jones says hello, Schwartz smells a sharp odor, like fruit gone bad, on her breath. It suggests acid buildup in her body, ...Read more

'Breaking a promise': California deficit could halt raises for disability workers
SACRAMENTO, Calif. — Families of people with intellectual and developmental disabilities say Gov. Gavin Newsom is reneging on a scheduled raise for the workers who care for their loved ones, and advocates warn of potential lawsuits if disability services become harder to get.
Citing California’s budget deficit, the Democratic governor wants...Read more

Steward Health goes bankrupt after mounting financial trouble
Steward Health Care System LLC filed for bankruptcy early Monday after a period of mounting financial challenges and government scrutiny over the impact of its hospital closures on patients.
The Dallas-based firm is finalizing a rescue loan with landlord Medical Properties Trust Inc., according to the filing. It sought Chapter 11 protection in...Read more

How does the brain think?
Curious Kids is a series for children of all ages. If you have a question you’d like an expert to answer, send it to curiouskidsus@theconversation.com.
How does the brain think? – Tom, age 16, San Diego, California
Have you ever wondered how your brain creates thoughts or why something randomly popped into your head? It ...Read more
Mayo Clinic Q&A: Using radiation therapy to treat brain tumors
DEAR MAYO CLINIC: I have been diagnosed with a brain tumor and advised to have radiation therapy. I'm very nervous about this and the risks for me, my family and, potentially, another cancer diagnosis. Can you explain why radiation therapy could be right for me?
ANSWER: Radiation therapy has been used to treat cancer since the beginning of the ...Read more

Is emotional eating sabotaging your weight-loss efforts?
You know losing that extra weight would be good for your health. Your health care team talked with you about how obesity increases your risk of other health issues, such as heart disease, diabetes, high blood pressure and certain types of cancer. They even worked out a healthy eating plan, and you want to stick to it. And you do well for many ...Read more

You've covered your copayment; now brace yourself for the 'facility fee'
Even if you have health insurance, you might expect to be charged a copayment for some routine care, like office-based exams and consultations. But you probably don’t expect to receive a bill a few weeks later charging you an extra $100 or more.
That’s the situation an increasing number of state lawmakers are looking to change. In ...Read more

Fact check: Robert F. Kennedy Jr. is wrong about a ban on NIH research about mass shootings
“Congress prohibits the NIH from researching the cause of mass shootings.”
Robert F. Kennedy Jr. in an April 21 post on X
____
The National Institutes of Health is the federal government’s main agency for supporting medical research. Is it barred from researching mass shootings? That’s what presidential candidate Robert F. Kennedy Jr....Read more

Ask the Pediatrician: Why adolescents need one-on-one time with their doctor
Aside from all the physical changes, adolescents are developing skills that help prepare them for adulthood. Part of this means participating more in their own health care. The adolescent well-child visits are tailored to support your family in this process.
Although pediatricians lend their own style to their examinations, many start the ...Read more
Popular Stories
- Mayo Clinic Q & A: Going plant-based: Exploring the myths, health benefits
- Lung cancer is the deadliest of all cancers, and screening could save many lives − if more people could access it
- One day in the frantic life of a children's emergency room nurse
- How does the brain think?
- California is investing $500M in therapy apps for youth. Advocates fear it won't pay off





