Health Advice
/Health

Bill of the Month: Sign here? Financial agreements may leave doctors in the driver's seat
Cass Smith-Collins jumped through hoops to get the surgery that would match his chest to his gender.
Living in Las Vegas and then 50, he finally felt safe enough to come out as a transgender man. He had his wife’s support and a doctor’s letter showing he had a long history of gender dysphoria, the psychological distress felt when one’s ...Read more

Could better inhalers help patients, and the planet?
Miguel Divo, a lung specialist at Brigham and Women’s Hospital in Boston, sits in an exam room across from Joel Rubinstein, who has asthma. Rubinstein, a retired psychiatrist, is about to get a checkup and hear a surprising pitch — for the planet, as well as his health.
Divo explains that boot-shaped inhalers, which represent nearly 90% of ...Read more

Should you be worried about bird flu? Here are 5 things to know about the virus
As a new virus takes center stage at the heart of a global outbreak, it’s easy to get flashbacks of March 2020.
Now more than four years after the world was rocked by a pandemic, H5N1, or avian or bird flu, has exploded in bird and livestock populations, and at least one human case has been confirmed by health officials.
This isn’t the ...Read more

A Pennsylvania study on racial bias in health care algorithms shows 'race is a bad variable'
PHILADELPHIA — University of Pennsylvania researchers undertook the most comprehensive review yet on the algorithms that doctors and health systems use to help make decisions on patient care and whether they contribute to long-standing racial biases in health care.
Their wide-ranging review of more than five dozen studies on algorithms and ...Read more
New COVID 'FLiRT' variants are spreading nationwide. Chicago health experts urge up to date vaccination
CHICAGO — A new family of COVID variants nicknamed “FLiRT” is spreading across the country, as vaccination rates in Chicago — as well as nationwide — remain concerningly low for some public health experts.
While symptoms and severity seem to be about the same as previous COVID strains, the new FLiRT variants appear to be more ...Read more

Amgen plows ahead with costly, highly toxic cancer dosing despite FDA challenge
When doctors began using the drug sotorasib in 2021 with high expectations for its innovative approach to attacking lung cancer, retired medical technician Don Crosslin was an early beneficiary. Crosslin started the drug that July. His tumors shrank, then stabilized.
But while the drug has helped keep him alive, its side effects have gradually ...Read more

A mother's loss launches a global effort to fight antibiotic resistance
LOS ANGELES -- In November 2017, days after her daughter Mallory Smith died from a drug-resistant infection at the age of 25, Diane Shader Smith typed a password into Mallory's laptop.
Her daughter gave it to her before undergoing double-lung transplant surgery, with instructions to share any writing that could help others if she didn't survive...Read more

Hospitals no longer must report COVID data to CDC, spotlighting clunky system
As COVID-19 swept over Phoebe Putney Health System and Southwest Georgia in the first days of the pandemic, residents and public health officials were desperate to know what patients had and how far it was spreading. The Albany, Georgia-based outbreak turned out to be one of the four largest in the world in terms of the percentage of the ...Read more
Counterfeit Botox found in California. Here's how to spot fake products and report them
Counterfeit versions of Botox have been found in multiple states, including California, according to the California Department of Public Health.
The health agency is warning consumers and medical providers to be wary of the counterfeit products, which have caused “hospitalizations and other serious reactions in people who received injections ...Read more

Bird flu has spread to cows in Colorado. Is avian influenza threat? We answer your questions.
DENVER — The bird flu has arrived in Colorado, and while it doesn’t represent anywhere near the threat that COVID-19 did at the beginning of the pandemic, people should take some precautions with animals that could carry it, experts say.
The Denver Post compiled the latest information on H5N1 highly pathogenic avian influenza, the bird flu ...Read more

Arizona’s now-repealed abortion ban serves as a cautionary tale for reproductive health care across the US
When the Arizona Supreme Court ruled on April 9, 2024, that the state’s Civil War-era law banning nearly all abortions was enforceable, it brought into stark reality the potential impacts of leaving reproductive rights up to the states to regulate, and the related consequences for women’s health.
The ruling, set to go into effect ...Read more

Environmental Nutrition: Lime time!
Tart and tangy, limes are the key ingredient that make flavors pop in our favorite sweet and savory foods.
The folklore
This tiny citrus fruit can certainly steal the spotlight as the star in tasty key lime pie, limeade and margaritas, but it shines even brighter in supporting roles, lending brightness and zing as condiment, garnish or finish....Read more

6 healthy summer foods to add to your diet
It’s almost summer — that amazing time of year when fresh produce abounds. We love that there’s an abundance of fresh, delicious and healthy choices. Better yet, many of summer’s fruits and vegetables are brimming with secret health benefits. Here are some of our favorites and why they're a particularly good choice in the summer.
1. ...Read more

Mayo Clinic Q&A: How can hospice care provide comfort to those with terminal illnesses?
DEAR MAYO CLINIC: I have a loved one who was referred for hospice care. I’m not sure what this means. Can you share more about what hospice care is and how it may help my family member?
ANSWER: Hospice care might be an option for people who are nearing the end of life due to a terminal illness and have exhausted all other treatment options.
...Read more
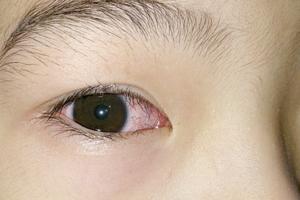
Mayo Clinic Q & A: Seeing clearly: Understanding pink eye
DEAR MAYO CLINIC: My entire family was recently affected by pink eye, including my mom, my nephews, my sister and even her dog! How is it so contagious? And what is it?
ANSWER: Pink eye is an inflammation of the lining of the eyelid and eyeball. The medical term for pink eye is conjunctivitis. Just as its name, pink eye, tells us, the most ...Read more

Mayo Clinic Q&A: Managing migraine: What's right for me?
DEAR MAYO CLINIC: I've dealt with migraine attacks for years. So has my mom. I typically manage by using over-the-counter pain medication that includes caffeine as soon as my symptoms begin. Then, if my migraine doesn't improve, I use a prescription triptan medication. But my headache days have been increasing, and my doctor suggested I try a ...Read more

Do dying people have a 'right to try' magic mushrooms? 9th Circuit weighs case
Do dying patients have a "right to try" illegal drugs such as psilocybin and MDMA if they might alleviate end-of-life suffering from anxiety and depression?
That question is now before one of the nation's highest courts, with a Seattle-based palliative care physician appealing a U.S. Drug Enforcement Administration decision barring him from ...Read more

California is investing $500M in therapy apps for youth. Advocates fear it won't pay off
With little pomp, California launched two apps at the start of the year offering free behavioral health services to youths to help them cope with everything from living with anxiety to body acceptance.
Through their phones, young people and some caregivers can meet BrightLife Kids and Soluna coaches, some who specialize in peer support or ...Read more

Bird flu is bad for poultry and dairy cows. It's not a dire threat for most of us -- yet
Headlines are flying after the Department of Agriculture confirmed that the H5N1 bird flu virus has infected dairy cows around the country. Tests have detected the virus among cattle in nine states, mainly in Texas and New Mexico, and most recently in Colorado, said Nirav Shah, principal deputy director at the Centers for Disease Control and ...Read more
Lung cancer is the deadliest of all cancers, and screening could save many lives − if more people could access it
Many medical organizations have been recommending lung cancer screening for decades for those at high risk of developing the disease.
But in 2022, less than 6% of people in the U.S. eligible for screening actually got screened. Compared with other common cancer screenings, lung cancer screening rates fall terribly behind. For ...Read more
Popular Stories
- New COVID 'FLiRT' variants are spreading nationwide. Chicago health experts urge up to date vaccination
- A Pennsylvania study on racial bias in health care algorithms shows 'race is a bad variable'
- A mother's loss launches a global effort to fight antibiotic resistance
- Amgen plows ahead with costly, highly toxic cancer dosing despite FDA challenge
- Should you be worried about bird flu? Here are 5 things to know about the virus





