Health
/ArcaMax

Which Cities Consume The Most Marijuana
It’s popular – but where is it the most and least consumed – here are the cities and how they rank with marijuana Recreational weed is in legal in 24 states, DC and some territories. Over 50% of their country has access to gummies, vapes and special chocolates without breaking the law. Thirty-eight states have medical marijuana, which the...Read more

All The Ways To Consume Cannabis
Unlike alcohol – cannabis can be consumed in a ton of different ways – here is a quick overview Marijuana has become mainstream and popular with over 85% of population believing it should be legal in some form. One of the things as weed “comes out the closet” is a the entry of a bunch of new users and former “closet smokers”. These ...Read more

Can CBD Help With An Overactive Bladder
Having to “go” frequently is an issue which causes multiple problems….can CBD help?
From watching a movie, going to live sports or performances to car trip, the frequent need to go can disrupt day to day life in big and little ways. Over 33 million Canadians and Americans live with overactive bladder disease (OAB). While not as life-...Read more

Marijuana Dosing For The First Half Of The Week
Monday through Wednesday tends to be a bear before the sweet promise of the weekend – but how to chill without a hangover or adding extra to the belly.
It was common and part of the American myth, guys would come home after work and pop a beer. Or stop by their “local” for a quick one before heading to the house. But the world has ...Read more

Toast 4/20 With A THC Infused Mocktail
Want to celebrate 4/20 with friends out and about – what about toasting with a THC infused mocktail?
It is the high holiday of marijuana use. You want to go out and have some fun, so why not toast 4/20 with a THC infused mocktail? Friday and Saturday are the busiest night at bars and sometimes you want something other than a vape or a ...Read more

These 5 Woman Are Changing 4/20
The marijuana industry is still driven by men – but these 5 woman are changing 4/20 and upending the industry by focusing on the future. Since it started becoming legal, the marijuana industry has been filled with bros trying to run everything. Some smart, some shysters – they have had an oversized say in the direction of where legal ...Read more

The Best Tips On How To Avoid Weekend Couchlock
Nothing like kicking back on the weekend – but there is a fine line between chilling and couchlock
Many a Saturday has been spoiled by too much fun on a Friday night. Hangovers have been a weekend curse for generations. While it is fun to let loose, if you go to far it can spoil part of the weekend. With marijuana mainstreaming, it is ...Read more

Vinyls and Marijuana Go Together
4/20 is also National Record Store Day – can there be a better combination?
What is old is new again, records stores are making a comeback. Record Store Day shares a celebration day with cannabis community. Like 4/20, it involved by a bunch of afficandios lifting up independent retailers and raising awareness and celebrating the multi-...Read more

Are You Really Ready To Try THC-P
THC-P is not for beginners – it is for the consumer who is well versed in the journey – and managing a high.
The high holiday of 4/20 is right around the corner and marijuana aficionados are thinking about celebrating. Some will go all out, other will chill and relax. It is a major holiday for dispensaries and a great time to experiment ...Read more

Will House Speaker Chaos Hurt Cannabis Again
The House seems to be about ready to have speaker chaos again. Will it hurt the cannabis industry like last time? It seems the US House of Representative might be thrown into turmoil again. Last fall, rejection of then House Speaker Kevin McCarthy lead to a bruising fight for a new leader. The winner of the fight is Rep. Mike Johnson, who is...Read more
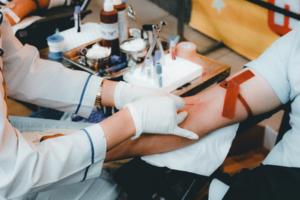
Can Marijuana Consumers Donate Blood
Blood donations are down, but the need is up. More people are needed to “open up” – but can you if you consume marijuana?
Every five seconds, at least two hospital residents need blood transfusions, Charles Richard Drew, an African-American surgeon and medical researcher helped create them, developing improved techniques for blood ...Read more

5 Things To Keep In Mind When Using Cannabis For Sleep
Cannabis and sleep go together like pillows and blankets. Here’s a few things you should keep in mind when using the herb to help you rest. The Fresh Toast – Medications have side effects, so what about something natural. Here are 5 things to keep in mind when using cannabis for sleep
Despite being used as a sleep aid, NyQuil is not one. ...Read more

Is Cannabis Good For The Planet
As Earth Day approaches, is growing cannabis good for the planet – it is a bit of a mixed bag As Earth Day approaches, everyone is evaluating what is good for the planet. Plastic, deforestation, fake fur, and Temu’s fast fashion are clearly bad for the planet, but others are more nuanced. Ever since Morton Hilbert started the movement, ...Read more

What Is THC
Everyone has heard of weed, marijuana, cannabis and more – but do you know exactly what is THC, what is does and is it in CBD? Marijuana and CBD have become mainstream and popular over the last few years. And mainstream companies from Constellation to Miracle Grow have become part of the industry. The American Medical Association have back ...Read more

Choosing The Right Edible
Edibles have become very popular – finding the right one for you is key! With over 50% of the country’s population having access to recreational cannabis, it has gone mainstream. Over 85% of the population believe it should be legal in some form and now there is a whole movement around California sober. As access to legal marijuana ...Read more

Why Marijuana Makes You Laugh
Laughter can be turn a bad situation around and make you feel a whole lot better. Here is why marijuana make you laugh. Laughter may not be the best medicine – but it is probably in the top three. Norwegian researchers conducted a 15-year study on the link between sense of humor and mortality among 53,556 women and men in their country. ...Read more

Cannabis Industry Employs The Same As These Companies
The cannabis industry is a growing industry with people who are passionate about their job
The cannabis industry has had a rough couple of years, but things are looking brighter. The one constant positive is consumer demand has continuously increased. You know it is good when Missouri has over $1 billion in sales last year. And, despite the...Read more

Can CBD Help Severe Weather Anxiety
One in 10 have severe weather anxiety – can CBD help people cope?
Severe weather has been on the rise for the last 10+ years, and for the one in ten who have storm anxiety, it is making things worse. Science has noted events including extreme precipitation, droughts and forest fires have become more frequent. Signs of storm anxiety include ...Read more

What Is Ice Water Extraction And Some DIY Tips
Not for the casual user, ice water extraction is when you really need some down, chill time.
Like wine, whiskey, tequila and a few other things, there are casual users and serious, serious users. A concentrate is for the experienced user who can manage a bit of a ride. So what is ice water extraction? It is a form of a concentrate which ...Read more

THC Has More Benefits Than Just Getting You High
People love THC for the fun – but if you look past it, there are other benefits Marijuana is becoming increasing popular and over 85% believe it should be legal in some form. Unlike alcohol, cannabis has proven medical benefits. Mentally, people recognize it as a help with PTSD, pain, chemotherapy, and more, but generally people see it as a...Read more





